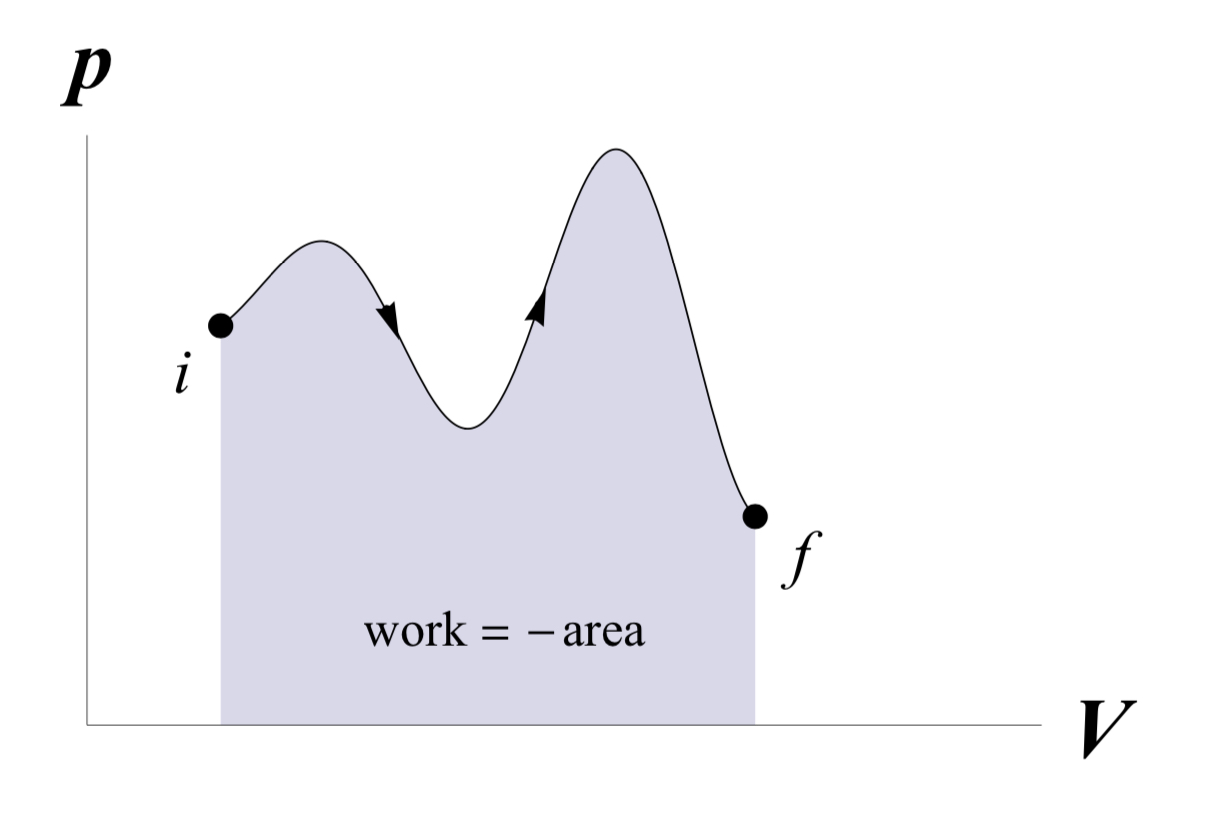
Work done by a gas during a quasi-static volume change
A volume change, i.e. the contraction or expansion of a container, is a mechanical change — and therefore any energy transferred due to this process will be work. The amount of work due to an infinitesimal quasistatic volume change is
where
Derivation
Consider a container of an ideal gas expanding in one direction. The gas therefore exerts a force
due to pressure, resulting in a displacement ⃗ 𝐅 in that one direction. The total volume change will be ⃗ 𝐬 , where ⃗ 𝐀 ⋅ ⃗ 𝐬 is the normal area vector: ⃗ 𝐀 ⃗ 𝐅 = 𝑝 ⃗ 𝐀 đ 𝑊 = − ⃗ 𝐅 ⋅ 𝑑 ⃗ 𝐬 = − 𝑝 ⃗ 𝐀 ⋅ 𝑑 ⃗ 𝐬 = − 𝑝 𝑑 𝑉